IVC Filter Lawsuit: What Patients Need to Know About Risks, Injuries, and Legal Action
Thousands of patients across the United States have filed an IVC filter lawsuit after suffering serious complications from implanted blood clot filters. These devices, intended to prevent pulmonary embolism, have been linked to injuries such as filter fracture, migration, and perforation of the vena cava. Many of the cases involve retrievable IVC filters made by companies like Cook Medical, Bard, and Cordis. As more adverse events are reported and consolidated into multidistrict litigation, patients and families are seeking answers and legal compensation for their injuries.
In this blog, we explain how IVC filters work, outline the risks linked to their failure, review the legal history behind current lawsuits, and discuss who may qualify to file a claim with the help of an experienced mass tort attorney.
What Is an IVC Filter and Why Are They Implanted?
IVC filters are medical devices placed in the body to prevent life-threatening blood clots. These filters are associated with serious complications, which have led to a rise in IVC filter lawsuits across the United States.
Purpose of an IVC Filter
An inferior vena cava (IVC) filter is a small metal device implanted into the inferior vena cava, the large vein that carries blood from the lower body to the heart. The primary purpose of the filter is to catch blood clots before they reach the lungs, where they can cause a pulmonary embolism.
Doctors may recommend an IVC filter when a patient is at high risk of developing blood clots but cannot safely take blood thinners. This includes patients with deep vein thrombosis, trauma injuries, or recent surgeries.
Types of IVC Filters
There are two main types of vena cava filters: permanent filters and retrievable IVC filters. Permanent filters are designed to stay in the body indefinitely. Retrievable filters are meant to be removed once the risk of developing blood clots has decreased. However, many retrievable filters are not removed in a timely manner, which can lead to serious complications.
Common Manufacturers of IVC Filters
Several companies manufacture IVC filters, including Cook Medical, C.R. Bard, Cordis Corporation, Rex Medical, and Boston Scientific. These filters are often the subject of blood clot filter lawsuits due to reports of filter fracture, migration, and perforation of the vena cava. Models such as the Bard Recovery Filter, Celect Filter, Meridian Filter, and Cordis Optease Filter have been linked to adverse events.
FDA Concerns and Medical Use
The U.S. Food and Drug Administration (FDA) issued a safety communication in 2010 and again in 2014. These alerts recommended that doctors consider removing retrievable IVC filters as soon as the patient no longer needs protection from pulmonary embolism. Despite these warnings, thousands of patients still live with filters implanted for longer than necessary, increasing the risk of filter complications.
Documented IVC Filter Complications and Failures
Many IVC filter patients have experienced serious complications after the device was implanted. These adverse events have become the basis of ongoing IVC filter lawsuits and multidistrict litigation.
Device Fracture and Filter Migration
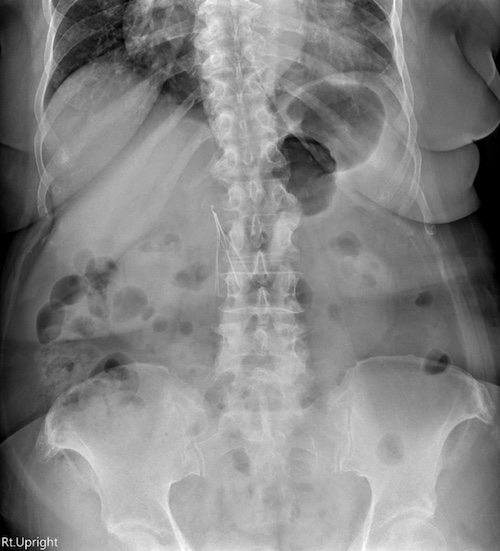
One of the most common issues reported is filter fracture. Parts of the IVC filter can break off and travel through the bloodstream. These fragments can lodge in the heart, lungs, or other organs, causing serious injury or requiring emergency surgery.
Filter migration is another known risk. The filter can move from its original position in the inferior vena cava and become lodged in another part of the body. Migration can result in pain, internal bleeding, or life-threatening damage to vital structures.
Perforation of the Vena Cava and Surrounding Organs
Retrievable filters that remain in the body too long can pierce the wall of the inferior vena cava. In some cases, the filter arms extend into nearby organs such as the spine, kidneys, or intestines. This can lead to severe complications, including internal bleeding and organ damage.
Studies published in JAMA Internal Medicine and other medical journals have confirmed high rates of filter penetration in certain models. Celect filters and Bard Recovery filters have been among the most frequently cited in these findings.
Retrieval Complications
Filters that are designed to be temporary often become embedded in the vein wall. This makes removal difficult and dangerous. In some cases, retrieval attempts have failed or caused more damage, such as tearing the vein or leaving fragments behind.
Many patients undergo multiple procedures to remove a filter that was supposed to be retrievable. These failed retrievals are often cited in blood clot filter lawsuits, especially where medical professionals were not warned of the risks by the manufacturer.
Adverse Event Reports and FDA Warnings
The FDA’s MAUDE database contains thousands of adverse event reports related to inferior vena cava filters. The agency issued safety communications in 2010 and 2014 to alert doctors about the importance of removing filters once the risk of pulmonary embolism passes.
Legal History and the Emergence of IVC Filter Lawsuits
The rise in IVC filter complications led injured patients to file lawsuits across the country. These cases allege that manufacturers failed to warn doctors and patients about known device risks.
Early Lawsuits and Legal Claims
The first IVC filter lawsuits were filed against companies like C.R. Bard and Cook Medical. Plaintiffs claimed that filters such as the Bard Recovery Filter and the Celect Filter were defectively designed and prone to fracture and migration. Many suits also accused manufacturers of failing to inform medical professionals about these dangers.
Claims include design defects, failure to warn, negligent misrepresentation, and breach of warranty. Injured patients sought compensation for medical expenses, lost wages, and pain caused by serious complications such as filter embolization and vena cava perforation.
Formation of Multidistrict Litigation
As the number of individual lawsuits grew, the Judicial Panel on Multidistrict Litigation (JPML) consolidated similar cases into multidistrict litigation, or MDL. This allowed courts to handle pretrial proceedings more efficiently.
The Cook IVC Filter MDL was created in 2014 and assigned to the Southern District of Indiana. It is overseen by Senior Judge Richard L. Young and Magistrate Judge Tim A. Baker. The Bard IVC Filter MDL was established in the District of Arizona in 2015. Both MDLs involve hundreds of IVC filter cases involving retrievable filters that allegedly failed after implantation.
Notable Manufacturers and Named Defendants
Cook Medical, C.R. Bard, and Cordis Corporation have faced the most lawsuits related to IVC filters. Lawsuits have focused on the Cook Celect filter, Bard G2 and Recovery filters, and Cordis Optease and TrapEase filters. Other companies named in legal claims include Boston Scientific and Rex Medical.
Each of these manufacturers is accused of selling devices that caused serious injury without providing proper warnings. Some IVC filter lawsuits also reference internal company documents that allegedly show early knowledge of failure rates.
Ongoing Litigation and MDL Status
Both the Cook and Bard MDLs remain active with pending dockets. Bellwether trials have already taken place to test the strength of claims. Some verdicts have resulted in compensation for plaintiffs, while others favored the defense. Settlements are ongoing in many cases, and more trials are scheduled.
Who Is Eligible to File an IVC Filter Lawsuit?
Not every IVC filter patient qualifies to file a lawsuit. Eligibility depends on the type of filter used, the complications experienced, and the timeline of those injuries.
Patients Who Suffered Filter Complications
Patients who had an inferior vena cava filter implanted and later suffered complications may be eligible to file an IVC filter lawsuit. Common qualifying injuries include device migration, filter fracture, filter embolization, and perforation of the vena cava or other organs.
Eligibility also extends to patients who experienced failed retrieval attempts. Retrievable filters that became embedded or broke during removal may support a legal claim. In some cases, additional surgeries were required, or fragments could not be removed at all.
Filters Made by Specific Manufacturers
Many legal claims involve IVC filters made by Cook Medical, C.R. Bard, and Cordis. If a patient received a Bard Recovery Filter, G2 Filter, Cook Celect Filter, Cordis Optease, or TrapEase Filter, they may meet the criteria for filing a blood clot filter lawsuit.
These products are frequently mentioned in multidistrict litigation and have been linked to a high rate of adverse events. Plaintiffs often argue that manufacturers failed to warn doctors about the risks of filter fracture and device migration.
Time Limits for Filing a Lawsuit
Each state has a statute of limitations that sets a deadline for filing a legal claim. This time limit usually begins when the patient first discovers the injury or should have reasonably known the filter caused harm. Missing this deadline can prevent a lawsuit from moving forward.
It is important to speak with a mass tort lawyer as soon as possible. An attorney can review medical records and determine whether the case falls within the allowed timeframe for filing.
Family Members of Deceased Patients
In cases where a patient died as a result of an IVC filter complication, surviving family members may be able to file a wrongful death claim. These claims may involve compensation for funeral expenses, loss of financial support, and emotional distress.
Contact an Experienced IVC Filter Lawsuit Attorney Today!
If you or someone you care about experienced serious complications from an IVC filter, including filter fracture, migration, or failed retrieval, you may be entitled to compensation. Our team at Rueb Stoller Daniel has the experience and focus needed to handle complex IVC filter lawsuits across the country.
Contact us at 1-866-CALL-RSD for a free consultation today!